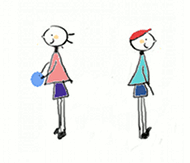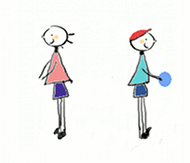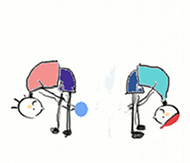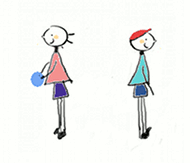Note: if a page name is blue, click it to visit that page. A word is blue, click it to visit the Vocabulary Page and see its definition.
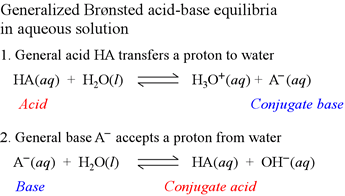
Remember the definitions you learned on the Properties, Reactions, and Definitions page? Well, one of them (known as the Brønsted-Lowry Theory) has a lot to do with conjugate acids and bases. The definitions stated that acids are proton (H+) donors, and bases are proton (H+) acceptors. After a base receives a proton, the remainder becomes a conjugate acid, and after an acid donates a proton, the remainder becomes a conjugate base.
Lets make things simple. Think of a proton as a ball being passed between two people like in the gif below. The person wearing red is an acid, and the person in blue is a base (just like on a pH scale). When the acid passes the ball overhead to the base, she is being a proton donor. When the base accepts it, he's being a proton acceptor. (However, they would not pass it below like in the gif)
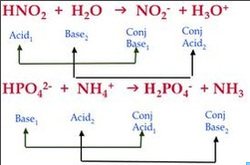
Now lets look at it in the form of an equation (see below)
HNO2 is an acid (you know that from the Indicators page), and H2O is a base. These are the reactants. On the right side of the arrow are the products, NO2- and H3O+. Notice that HNO2 no longer has a proton on the products side (it is now NO2-), because it donated it to the base. This makes NO2- a conjugate base. H2O received a proton and became H3O+, so it is the conjugate acid.
HNO2 is an acid (you know that from the Indicators page), and H2O is a base. These are the reactants. On the right side of the arrow are the products, NO2- and H3O+. Notice that HNO2 no longer has a proton on the products side (it is now NO2-), because it donated it to the base. This makes NO2- a conjugate base. H2O received a proton and became H3O+, so it is the conjugate acid.
Here's another example. Notice the "⇌" symbol, which means partial dissociation. This means that the acid/base is weak. (If you forgot about this, revisit the Strength, Buffers, and Concentration page)
Get it now? Good, because theres MORE!