Note: If a word is blue, click it to visit the Vocabulary Page and see its definition.
There are many different ways to identify if a solution is an acid or a base (using various indicators).
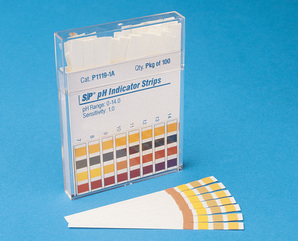
The most common is the pH Strip, in which you dip the strip in a liquid solution, and match the colors that appear with a category on the box.
There are many different ways to identify if a solution is an acid or a base (using various indicators).
The most common is the pH Strip, in which you dip the strip in a liquid solution, and match the colors that appear with a category on the box.
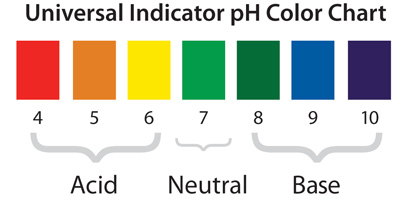
Other indicators include the Universal Indicator, which is a chemical that changes the color of an acid or base (see picture above). You match this color with one on the chart above, and then you will know if it is an acid or base. This is different from a pH strip because it also works on solids.
Phenolphthalein is also an indicator (see picture above). When combined with a base, it turns pink. If it's neutral (pH of 7), it's clear (useful for neutralization reactions).
Another common indicator is red litmus paper and blue litmus paper. In a base, litmus paper will turn to or stay blue. In an acid, it will turn to or stay red. (see picture below).
Phenolphthalein is also an indicator (see picture above). When combined with a base, it turns pink. If it's neutral (pH of 7), it's clear (useful for neutralization reactions).
Another common indicator is red litmus paper and blue litmus paper. In a base, litmus paper will turn to or stay blue. In an acid, it will turn to or stay red. (see picture below).
Sometimes you'll need to identify if something is an acid or base and you won't have it on hand or know its molarity. In this case, you can often use the molecular formula.
A strong acid's molecular formula will start with H, and a strong base's/alkali's formula will end with OH.
For example, HCl is hydrochloric acid, and NaOH is sodium hydroxide (an alkali).
A strong base will end with the word "oxide," and a strong alkali will end with the word "hydroxide."
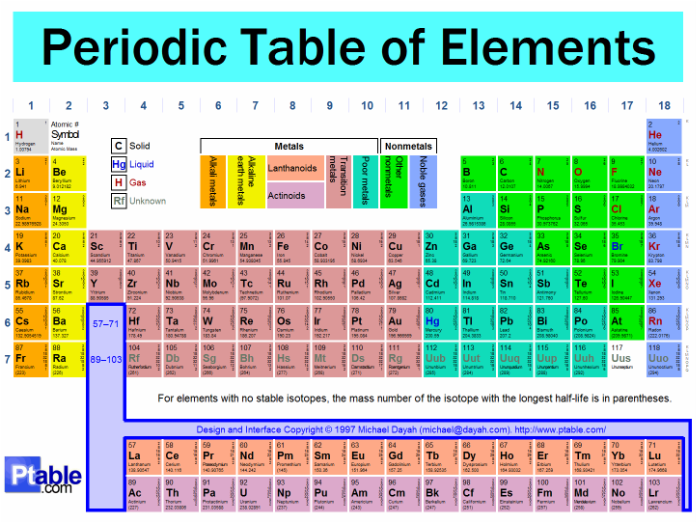
Also, strong bases/alkalis are metal oxides/hydroxides (if it dissolves in water its an alkali), so they are in group 1 and group 2 on the periodic table (these are the first two columns on the left)
Acids are non-metal oxides dissolved in water, which are on the right side of the periodic table in the green section (non metals).
A trick: If something is an acid and you can eat it (ex. Citric Acid is in lemons), its weak.
A strong acid's molecular formula will start with H, and a strong base's/alkali's formula will end with OH.
For example, HCl is hydrochloric acid, and NaOH is sodium hydroxide (an alkali).
A strong base will end with the word "oxide," and a strong alkali will end with the word "hydroxide."
Also, strong bases/alkalis are metal oxides/hydroxides (if it dissolves in water its an alkali), so they are in group 1 and group 2 on the periodic table (these are the first two columns on the left)
Acids are non-metal oxides dissolved in water, which are on the right side of the periodic table in the green section (non metals).
A trick: If something is an acid and you can eat it (ex. Citric Acid is in lemons), its weak.
You'll learn to calculate the pH of an acid, base, or alkali soon. For now, keep reading!