Note: If a word is pink, click it to visit the Vocabulary Page and see its definition.
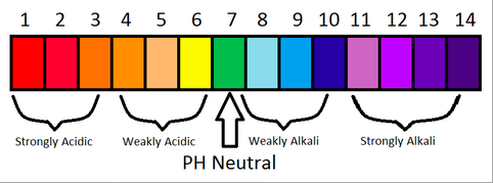
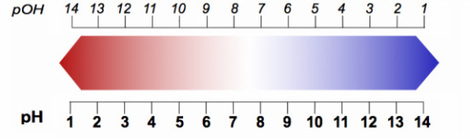
Strength is the ability to produce Hydrogen/Hydroxide ions. A strong acid is one that dissociates completely to form H+ ions, whereas a weaker acid only dissociates partially.
A strong alkali dissociates completely to form OH- ions, whereas a weak alkali dissociates partially.
A strong alkali dissociates completely to form OH- ions, whereas a weak alkali dissociates partially.
|
Complete Dissociation:
Hx (any generic acid) --> H+ |
Partial Dissociation:
Hx ⇌ H+ |
|
Complete Dissociation (Sulfuric Acid):
H2SO4 --> H+ + HSO4- |
Partial Dissociation (Acetic Acid):
CH3 + CO2H + H20 ⇌ CH3CO2- + H3O+ |
Examples:
|
|
A buffer is a solution that can resist pH change upon addition of an acid or base. It can neutralize small amounts of acids or bases, which keeps the pH level of the solution relatively stable. This is necessary for processes that require a stable pH level, such as blood (which maintains a pH of 7: Neutral). If a very strong acid or base were added to a buffer, it may change slightly, but for the most part any changes would be unnoticeable.
A buffer consists of a weak conjugate acid-base pair, which can mean one of two things: a weak acid and its conjugate base, or a weak base and its conjugate acid.
(You will learn about conjugate acids and bases a few pages from now)
A buffer consists of a weak conjugate acid-base pair, which can mean one of two things: a weak acid and its conjugate base, or a weak base and its conjugate acid.
(You will learn about conjugate acids and bases a few pages from now)
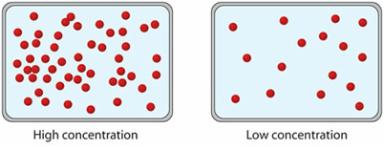
Concentration tells you the amount of substance per unit volume. Later in the website, you'll learn how to calculate the pH when you have the level of concentration.
Pretty straight forward, right?