Note: If a word is blue, click it to visit the Vocabulary Page and see its definition. If a page name is blue, click it to visit that page.
This page will teach you how to do the titration lab. The purpose of this experiment is to determine the concentration of a HCl solution using acid-base titration.
So what is titration? Titration is a technique used to find the concentration of a known solution. (If you've forgotten what concentration is, visit the Vocabulary Page)
If the concentration of an acid (like HCl) is unknown, you can use the known concentration of a hydroxide base to complete the experiment by using this neutralization reaction: Acid + Base --> Salt + Water
If the concentration of an acid (like HCl) is unknown, you can use the known concentration of a hydroxide base to complete the experiment by using this neutralization reaction: Acid + Base --> Salt + Water
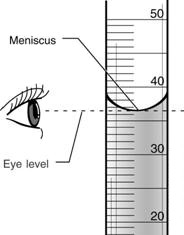
So let's get started on the experiment! But first, a caution: Hydrochloric Acid is a very strong acid and should not come in contact with your skin or eyes. Before doing the experiment, put on goggles and gloves, but your hair up, and make sure there is access to a shower just in case any chemicals touch your skin.
|
Materials:
To the right is a slideshow displaying the materials. |
|
|
Procedure:
|
Calculation Formulas:
- Moles = molarity x liters
- Molarity = moles / liters
- Moles of HCl = Moles of NaOH
- 1mL = .001L
Sample Calculation:
25.4mL of NaOH was used to neutralize 25mL of HCl
NaOH Concentration: .1 M
HCl Concentration: Unknown
25.4mL of NaOH was used to neutralize 25mL of HCl
NaOH Concentration: .1 M
HCl Concentration: Unknown
- NaOH : 25.4mL = .0254 L
- NaOH : .1 M x .0254 L = .00254 m
- The #moles of HCl will be the same as the #moles of NaOH, therefore the #moles of HCl will also be .00254 m
- HCl: 25mL = .025 L
- HCl: .00254 m / .025 L = .1016M
- The Concentration of HCl is therefore, .1016 M
YOU'RE DONE LEARNING! Now onto the quiz!